Overview
Olfactory perception begins by the recognition of odorants by a large repertoire of receptors in the sensory epithelium. Neurons expressing a given receptor are distributed in epithelium but project with precision to spatially invariant glomeruli in the olfactory bulb. Thus, odor is represented in a pattern of activated glomeruli. The activity of individual glomeruli is propagated to multiple third order olfactory areas including the anterior olfactory nucleus, cortical amygdala, lateral entorhinal cortex, olfactory tubercle, and piriform cortex. It remains largely unclear what function is served by these parallel outputs. Our research focuses on understanding olfactory processing in these third order areas and how sensory representation transforms in their outputs to downstream structures.
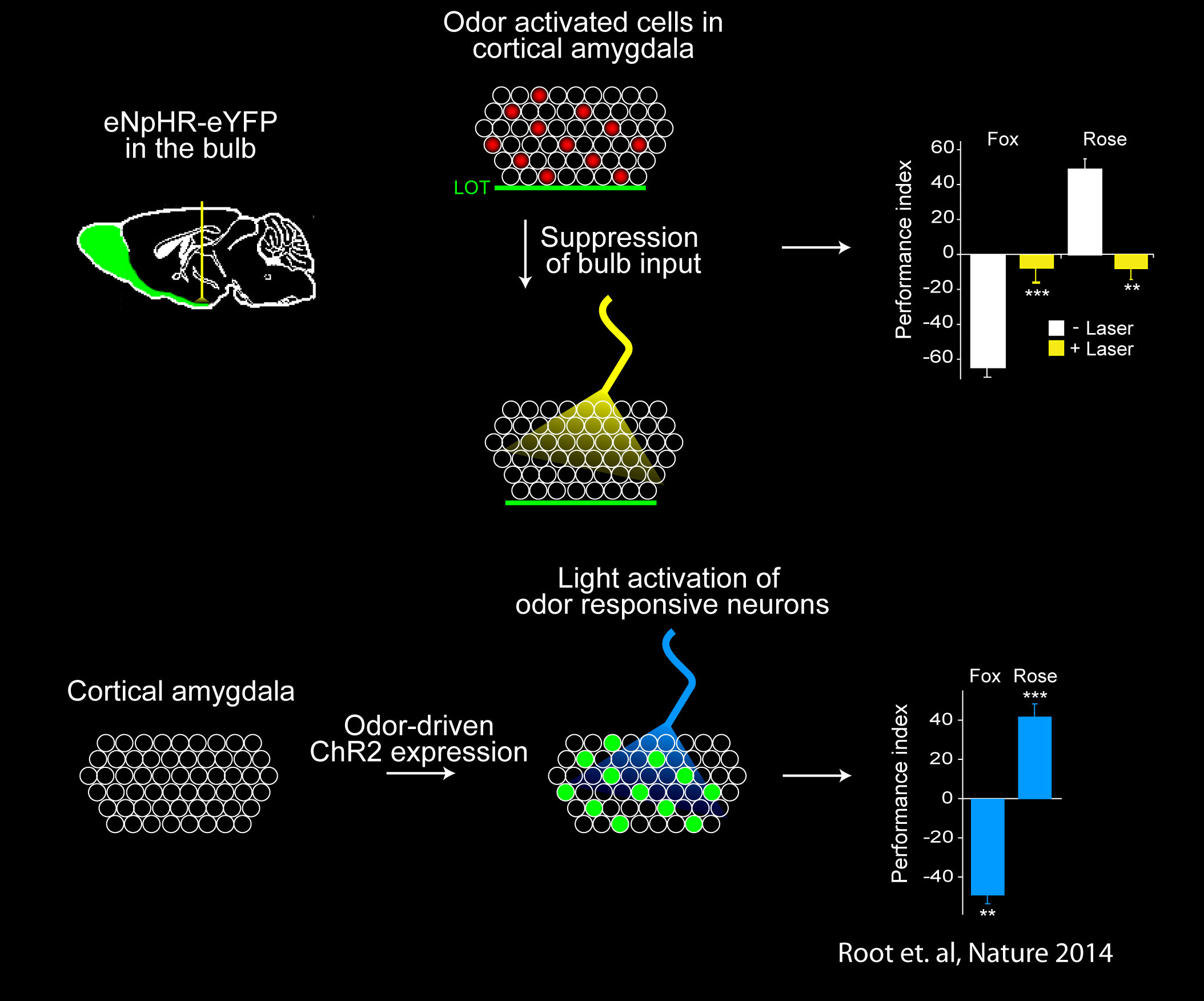
The cortical amygdala mediates innate olfactory behavior
Anatomic tracing experiments had implicated the cortical amygdala (plCoA) in genetically determined behaviors. We used optogenetic experiments to directly determine if the cortical amygdala is involved in innate odor-driven behavior. Indeed, we found that the stereotyped circuits from the olfactory bulb to the cortical amygdala are required for innate aversive and appetitive responses to odor. Moreover, we identified and manipulated the activity of these neurons by exploiting the promoter of the activity-dependent gene, arc, to express channelrhodopsin in neurons of the cortical amygdala activated by odors that elicit innate behaviors. Optical activation of these neurons leads to appropriate behaviors that recapitulate the responses to odors. Together these data demonstrate that the cortical amygdala contains distinct neurons that are necessary and sufficient for the generation of innate, odor-driven behaviors (Root et al., Nature 2014). The identification of neural circuits that elicit innate behaviors provides a foundation for dissecting sites of plasticity to afford behavioral flexibility.
Cell types and circuits of the cortical amygdala
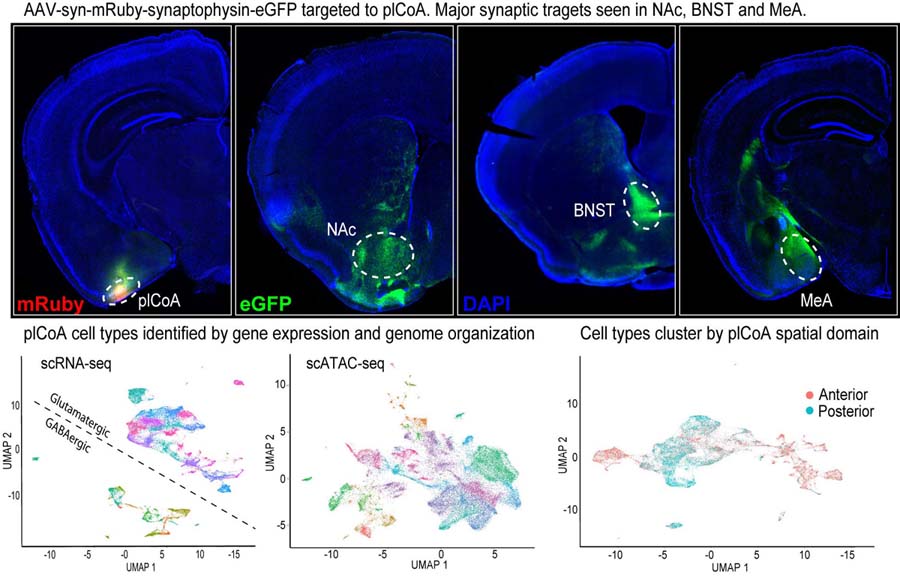
In ongoing work, we are identifying and manipulating the cell types and circuits of plCoA. Viral tracing experiments revealed that the major synaptic targets of the plCoA are the nucleus accumbens (NAc), BNST, and medial amygdala (MeA). Preliminary evidence indicates that the projections to the NAc and MeA mediate attraction and aversion, respectively. Further, we have used single cell genomics (scRNA-seq and scATACseq) to identify the cell types of the plCoA by gene expression and chromatin organization. Interestingly, there is a spatial gradient of cell types between different domains of plCoA suggesting a functional axis.
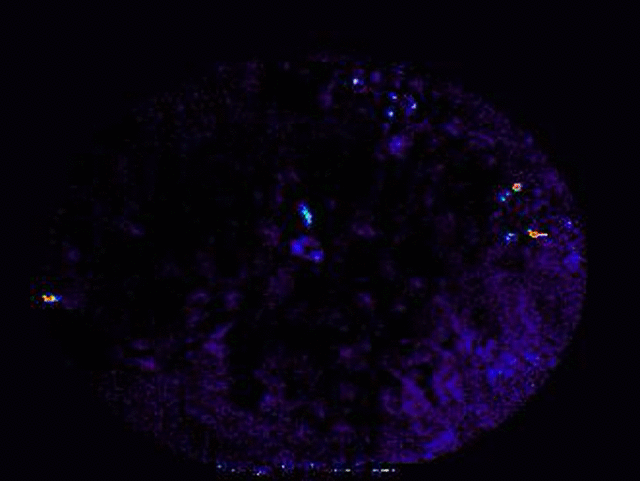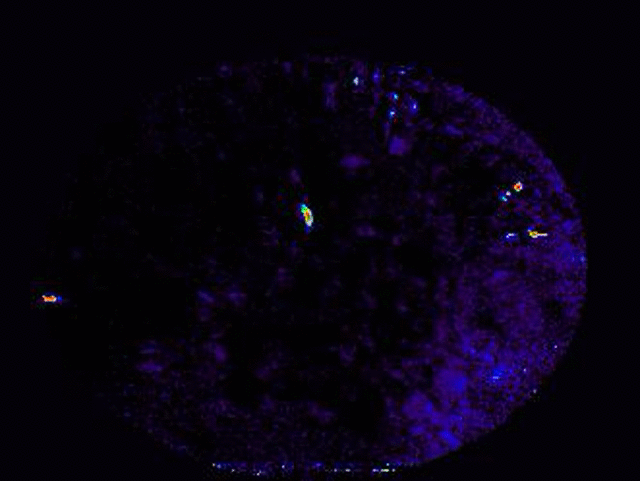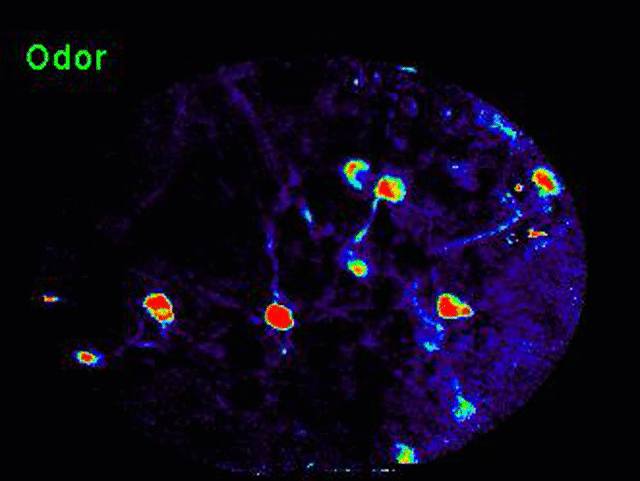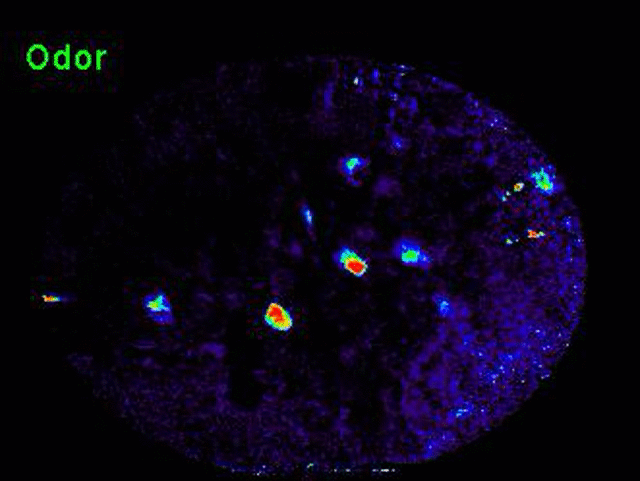
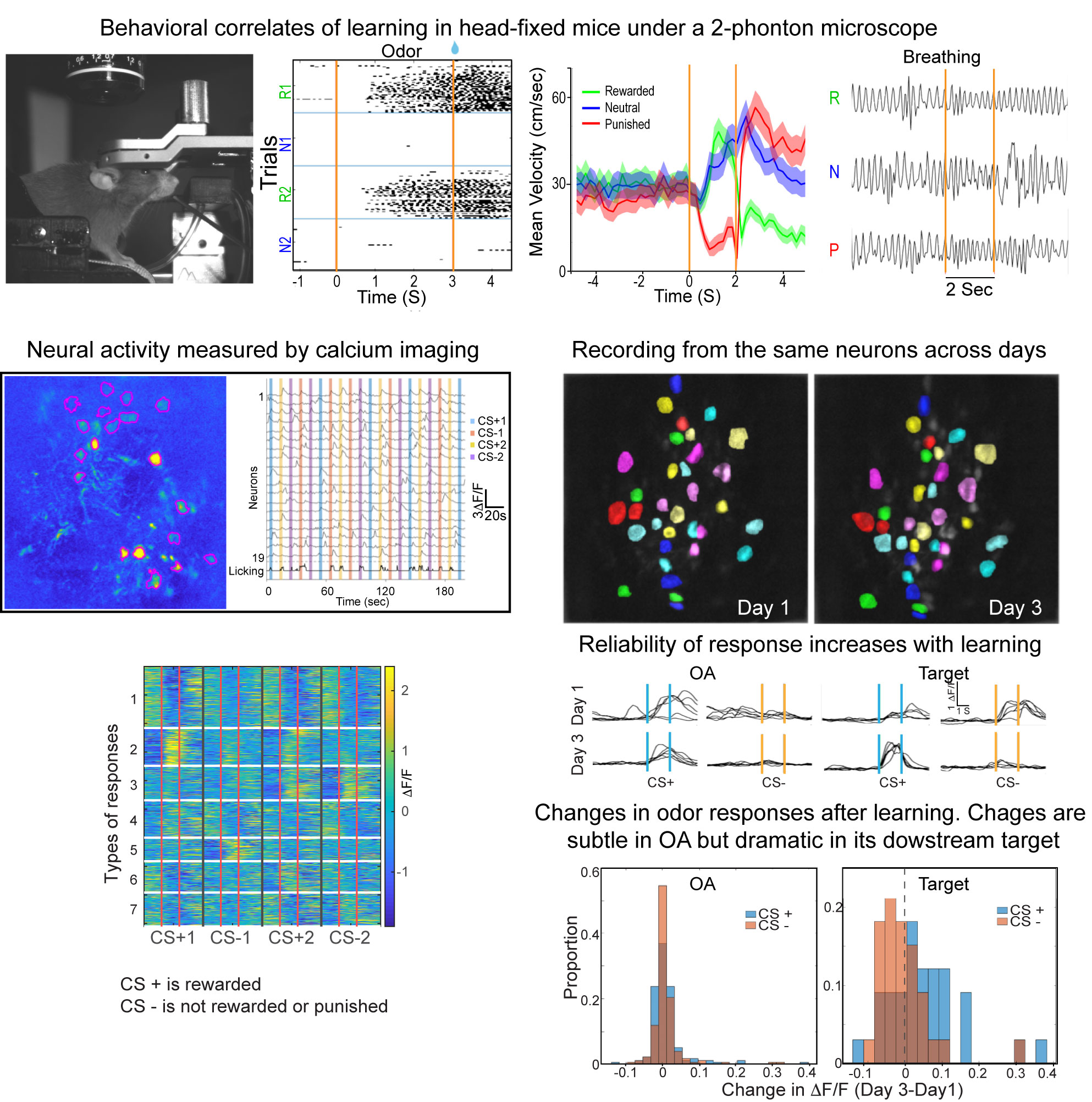
Transformation of odor representation between brain areas with experience.
We are using 2-photon endoscopic imaging to record neural activity in awake, behaving mice. We first established behavioral correlates of leaning in head-fixed animals whereby thirsty mice will lick a sipper for rewarded odors and pause walking behavior for punished odors; sniffing frequency does not change. Neural activity reveals a subtle change in odor representation in one third order olfactory brain area (OA), but the representation is dramatically transformed in its downstream target (target). How does this transformation occur? We are currently combining optogenetics and 2-photon imaging to optically dissect the functional connections between these brain areas.